- According to The World Stroke Organization, stroke is the second leading cause of death and disability worldwide.
- One of the most common effects of a stroke is hemiparesis, that is, the weakness of one half of the body.
- ReWalk Robotics Ltd has developed a robotic exosuit meant to aid in the rehabilitation of stroke victims.
First FDA-approved Exosuit for Stroke Rehabilitation
The ReWalk ReStoreTM is the first FDA-approved soft robotic exosuit for stroke rehabilitation.
A multicenter clinical trial was conducted to test the safety, feasibility, and, reliability of the device. 44 patients with post-stroke hemiparesis were recruited from rehabilitation centers across the US.
The trial aimed to improve the speed and gait of those suffering from post-stroke hemiparesis.
The ReStoreTM Exosuit – developed at Harvard University
The robotic exosuit consists of a lightweight waist pack and mechanical cables. The motor generates mechanical forces which are transmitted via cables to attachment points. One of the attachment points is located around the calf while another is present on the shoe insole.
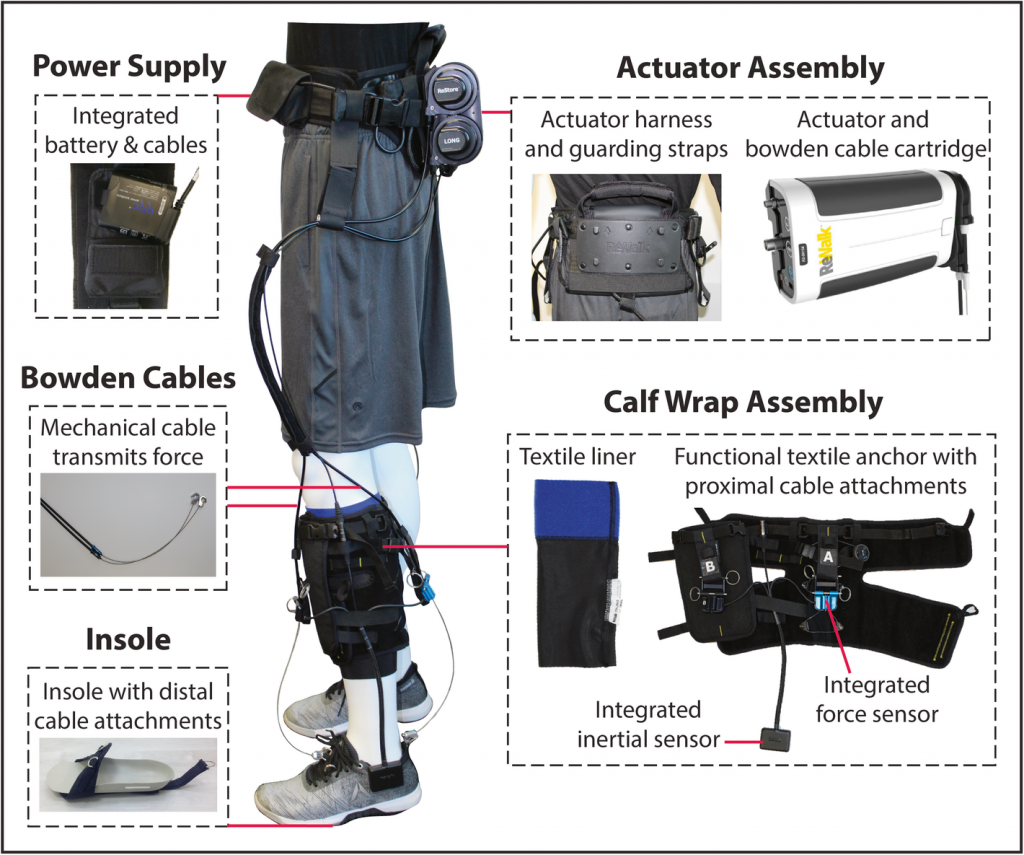
Source: Awad et al. Journal of NeuroEngineering and Rehabilitation
The device is designed to provide targeted assistance to patients during the plantar flexion and dorsiflexion phase of their gait. In simpler terms, it helps lift the patient’s affected leg in synchronized timing with their natural walking pattern.
By training the patient walking pattern in a more correct way, the expectation is that the brain will relearn and better restore the walking function lost after a stroke
Dr. Alberto Esquenazi
Additionally, real-time analytics from the device help physical therapists optimize a patient’s treatment and progress.
Post-Stroke Hemiparesis
Stroke is a leading cause of disability worldwide. According to estimates, 80% of stroke patients suffer from an impaired gait, likely due to hemiparesis. Post-stroke hemiparesis can result in impaired dorsiflexion and plantarflexion of the affected ankle, hindering mobility.
The trial focused on stroke patients that could walk 5 feet without any assistance. It required participants to complete 20 min of overground walking practice and 20 min of treadmill walking practice while receiving assistance from the device.
Findings of the trial
The findings were published in the Journal of NeuroEngineering and Rehabilitation.
We found that the ReStore provided targeted assistance for plantarflexion and dorsiflexion of the paretic ankle, improving the gait pattern
Dr. Nolan
After only 5 days of practice with the ReStoreTM, an increase was seen in the users’ assisted and unassisted walking speeds. Moreover, only a few device malfunctions were reported during the trial along with zero device-related falls.
The average satisfaction ratings for the device was between “quite satisfied” (i.e., 4) and “very satisfied” (i.e., 5).
“This is an important first step toward expanding options for rehabilitative care for the millions of individuals with mobility impairments caused by ischemic and hemorrhagic stroke.”
Dr. Nolan
The findings of the study thus demonstrate the exosuit could potentially play a key role in the rehabilitation of stroke patients.
Reference:
Awad, L.N., Esquenazi, A., Francisco, G.E. et al. The ReWalk ReStore™ soft robotic exosuit: a multi-site clinical trial of the safety, reliability, and feasibility of exosuit-augmented post-stroke gait rehabilitation. J NeuroEngineering Rehabil 17, 80 (2020). https://doi.org/10.1186/s12984-020-00702-5
Kim J, Thayabaranathan T, Donnan GA, et al. Global Stroke Statistics 2019 [published online ahead of print, 2020 Mar 9]. Int J Stroke. 2020;1747493020909545. doi:10.1177/1747493020909545
Lindsay MP, Norrving B, Sacco RL, et al. World Stroke Organization (WSO): Global Stroke Fact Sheet 2019. Int J Stroke. 2019;14(8):806-817. doi:10.1177/1747493019881353




