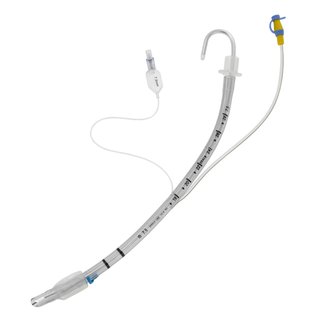
ASSET is the only multiport suction breathing tube for safe mechanical ventilation that has FDA approval. Moreover, it prevents the risks of ventilator-associated pneumonia. Ventilator-associated pneumonia is a very common infection. It happens when the material leaks into the breathing tube that enters the lungs.
Innovative Intervention
Aspire has come up with an innovative intervention for this challenge. Their device removes pathogenic subglottic fluid before it causes any infections. It is the first-ever and only multi-port subglottic suction ET tube. Furthermore, it has a tissue spacer and twenty-four ports that can remove pathogenic secretions and fluids without airway tissue blockage. Fluids becoming blocked due to airway tissue is the main problem with single-port devices.
Prevents Ventilator-Associated Pneumonia
When compared with other single-port ET suction tubes, the results are significantly better at removing fluid at every suction setting. The multiport system can allow subglottic drainage in multiple patient positions. In addition to the tissue spacer preventing blockage of suction and injury.
Dr Benjamin Wang said,
“Ventilator-associate pneumonia is the most costly infection in modern hospitals. Added to this, the rates of antibiotic-resistant infections are expected to increase in the coming years, which will make treating these complications more difficult and costly. Now is an opportune time to see how a proven innovation in airway management and infection control can help augment patient safety and cost reduction in all clinical settings. The COVID-19 pandemic has additionally meant that innovations in airway management are desperately needed. We are delighted to be joining the ABHI UK Pavilion to showcase how the Aspire can help tighten infection control, reduce costs, and enhance safety for patients across the globe.”
Subglottic drainage has proven to reduce the incidence of VAP, decreasing a patient’s hospital stay and mechanical ventilation duration. The ASPIRE ET tube accommodates different suction pressure ranges with the facility of hospital administration.